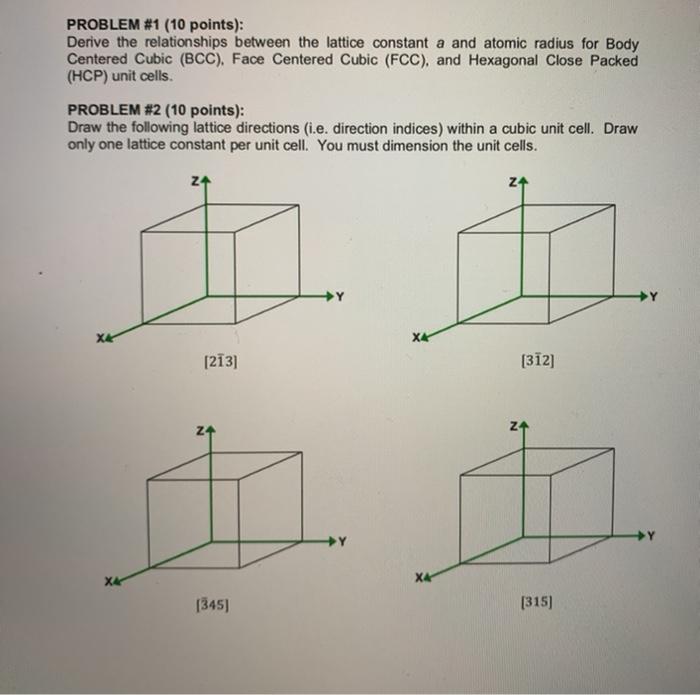
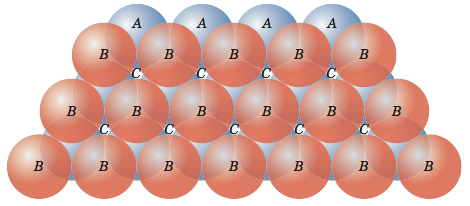
Unit Cell Chemistry, Atomic Radius, Density & Edge Length Calculations, Close Packed Structures - YouTube

SOLVED: Discussion QuEstions And PROBLEMS` The cesium chloride (CsCI) unit cell is similar to the body-centered cubic cell you built in Part B The center = sphere is taken to be a

Silver crystallises in a face - centred cubic in cell. The density of Ag is 10.5 g cm^-3 . Calculate the edge length of the unit cell.
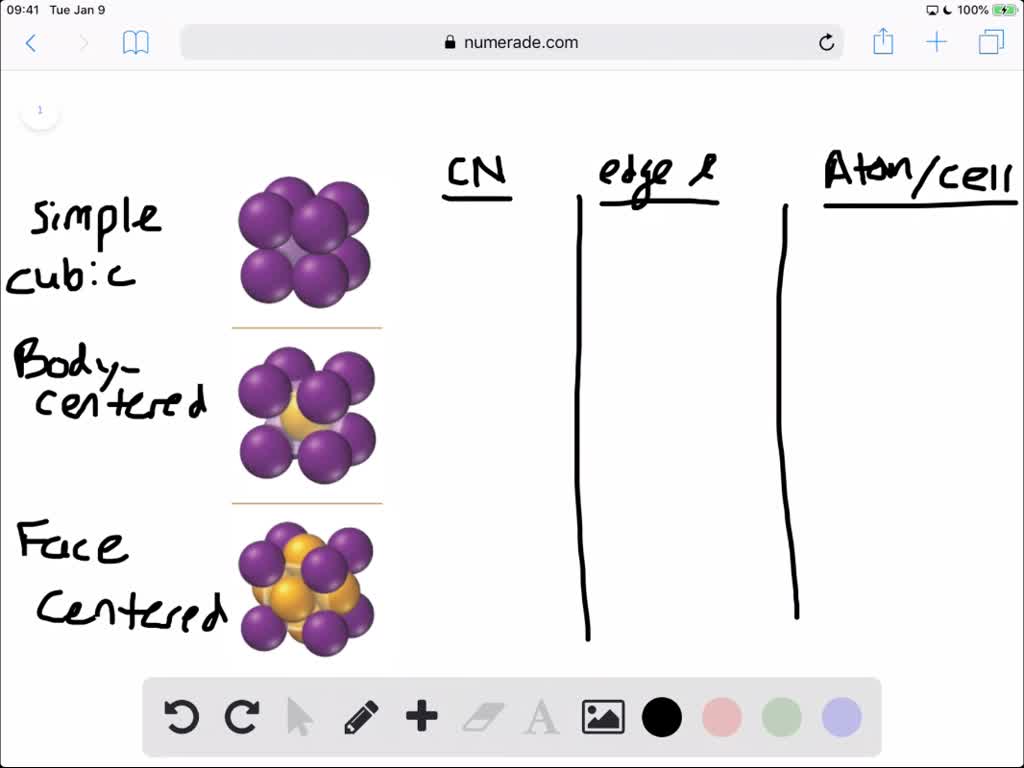
SOLVED:For each of the cubic cells in the previous problem, give the coordination number, edge length in terms of r, and number of atoms per unit cell.

Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu - YouTube

Unit Cell Chemistry, Atomic Radius, Density & Edge Length Calculations, Close Packed Structures - YouTube

Unit Cell Chemistry, Atomic Radius, Density & Edge Length Calculations, Close Packed Structures - YouTube
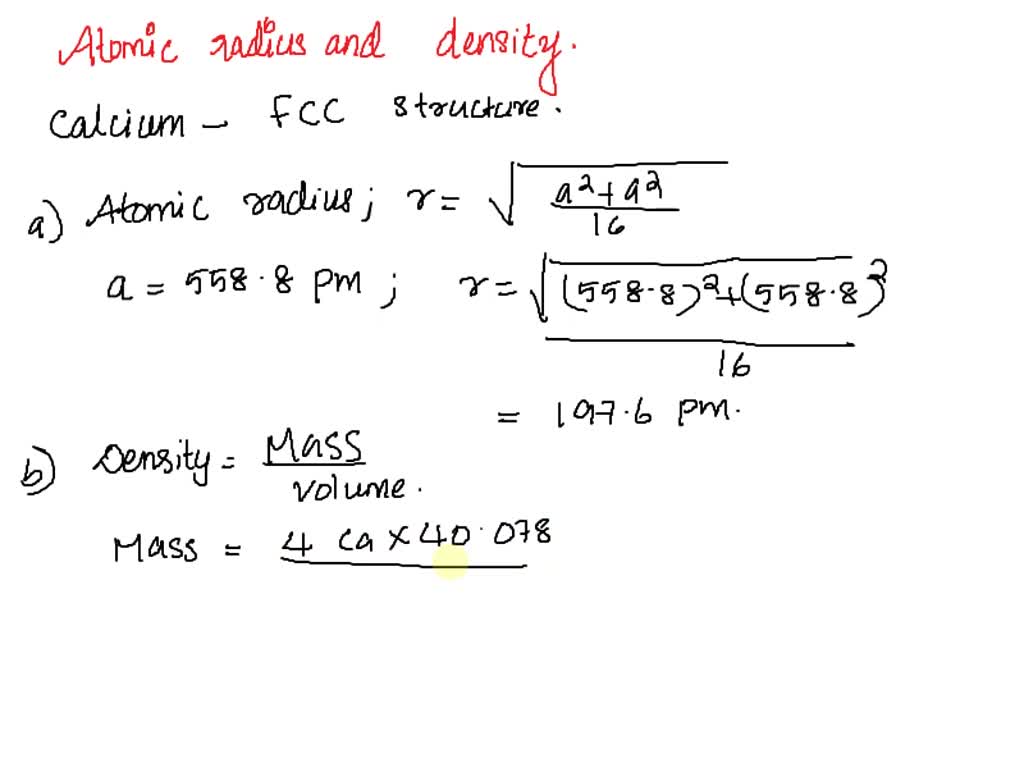
SOLVED: Calcium crystallizes in a face-centered cubic structure. The edge length of its unit cell is 558.8 pm.(a) What is the atomic radius of Ca in this structure?(b) Calculate the density of

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange

SOLVED: Determine the volume density of the atom in crystals with (a) simple -cubic,(b) body-centered cubic and(c) face-centered cubic crystal structures with a lattice constant a=5A.

An element has a body-centered cubic (bcc) structure with a cell edge of 288pm. The density...... - YouTube

Niobium crystallizes in body-centered cubic structure. If density is 8.55 g/cm3, Calculate...... - YouTube

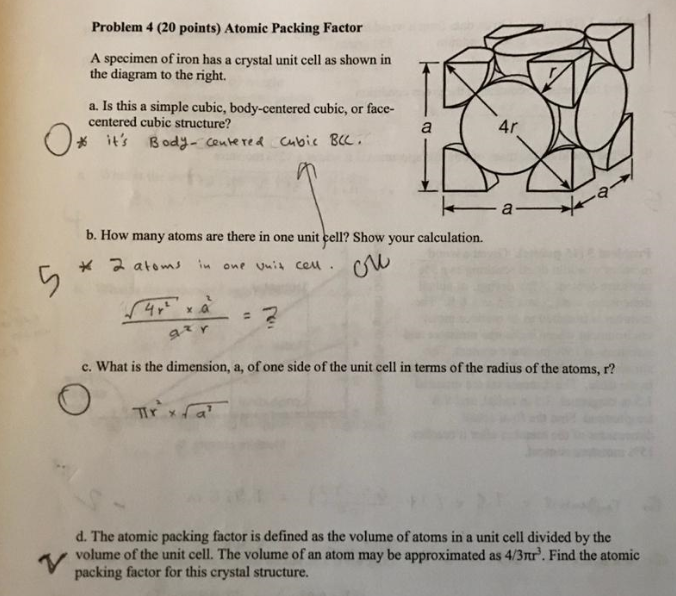
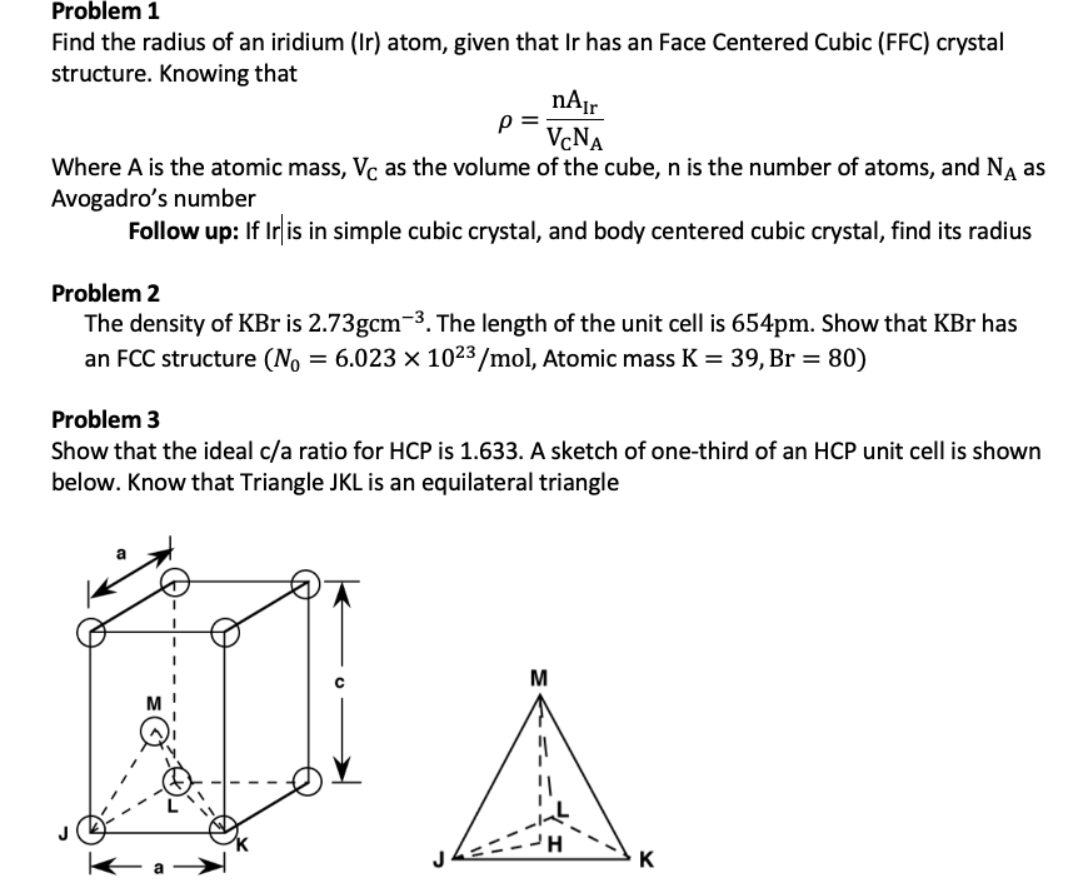
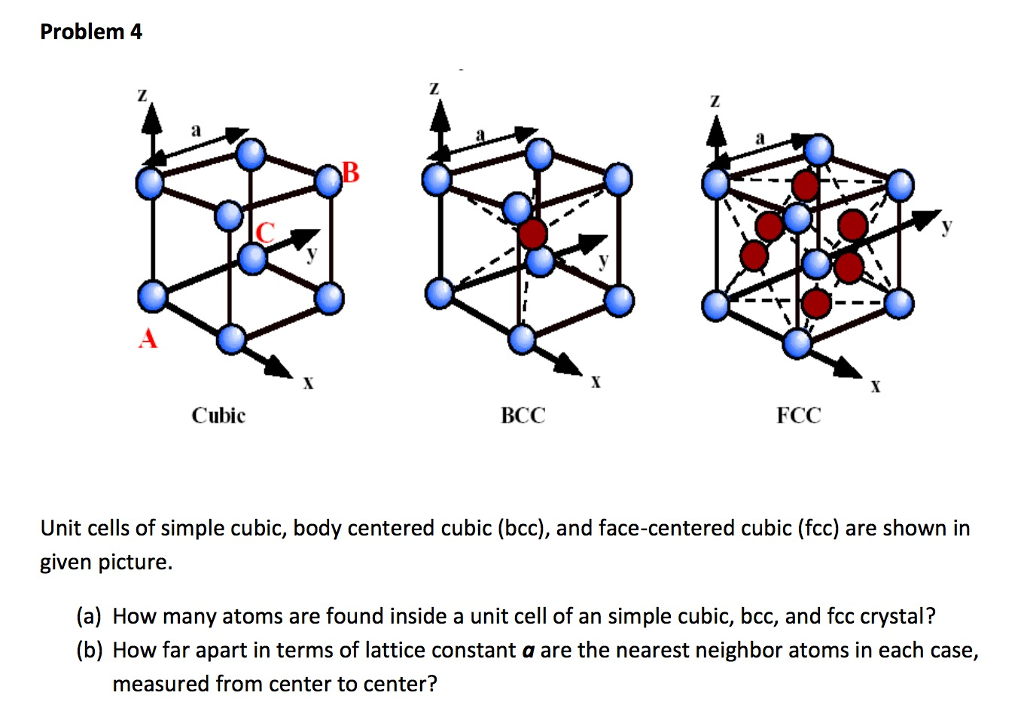
![Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com](https://media.cheggcdn.com/media/1eb/1eb9fa27-1dc7-4453-878c-a7f39a79cbf4/phppeImhN.png)